It’s important to choose a treatment plan based on PS, tumor characteristics, life expectancy, and comorbidities. In addition, factors such as risk stratification of the cancer and if the cancer is castration resistant or castration sensitive also need to be considered.1,2 Finally, patient preferences for treatment are taken into account to ensure optimal outcomes.
Consideration of current and past treatments, additional medications and side effects experienced are key to develop an optimal treatment plan for your patients now and in the future.
The potential for recurring side effects and/or repeat adverse reactions should be recognized and shared with your patients and their care partners.
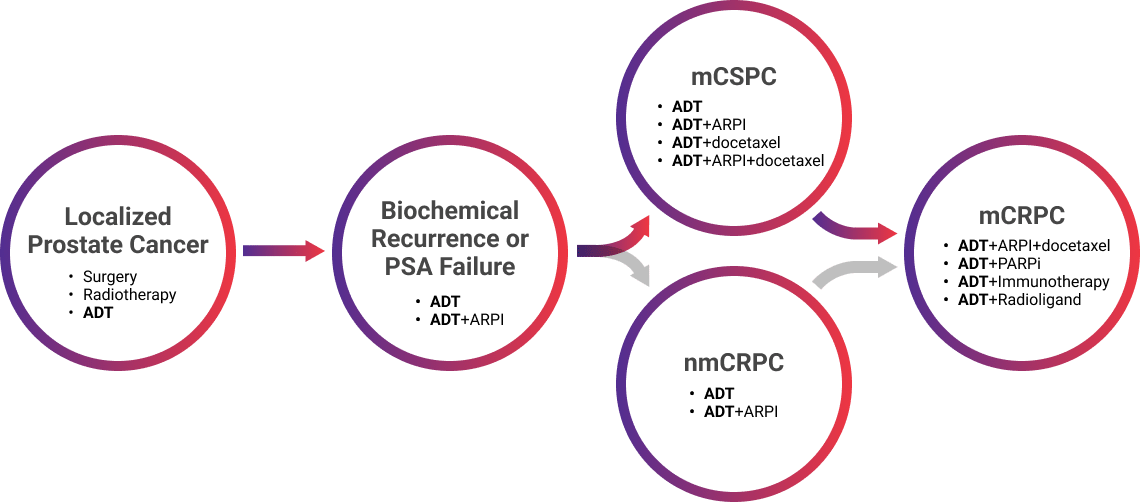
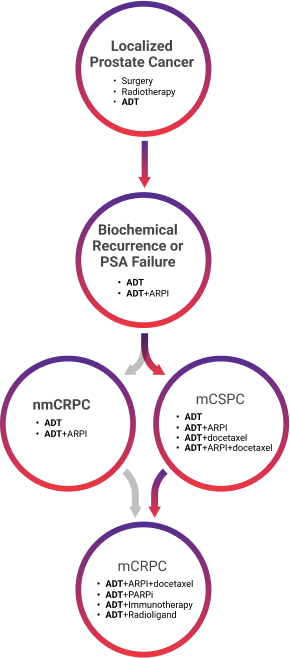
ADT is beneficial throughout the prostate cancer care continuum, as it can slow or stop growth in advanced prostate cancer by lowering testosterone (T) and provides a foundation for other treatments. Once a patient begins to progress, from localized to metastatic hormone-sensitive or -resistant prostate cancer, treatment options change, but ADT can be used regardless, as it is considered the foundation of care for locally advanced or advanced prostate cancer, as shown below.4,5
| Agonists5,8-13 | Antagonists5,8-13 | |
|---|---|---|
| Mechanism of action | Work by overstimulating the LHRH (also known as GnRH) receptors in the anterior pituitary gland, which desensitizes them over time. Continual binding of the agonists to the LHRH receptors results in an initial increase in LH release and T production, followed by a steep drop in both. Patients will reach clinically castrate levels of T in 2 to 4 weeks. | Work by blocking the GnRH (gonadotropin-releasing hormone) receptors in the pituitary gland, causing a faster decrease in LH and T, with castrate levels being reached in 2 to 3 days. |
| Benefits of initiating | Long-acting formulations provide patients with reliable, controlled suppression over time, which may help avoid periods of T breakthrough above castrate levels. | May benefit patients with spinal metastases due to the absence of initial T increase and others who require a rapid T suppression. |
| Route of delivery | Injected subcutaneously or intramuscularly. | Subcutaneous injection or oral. |
| Administration schedule | Depending on the product, can be administered monthly, every 3 or 4 months, or even just twice per year. | Taken once a day orally or injected every month. |
| Dosing flexibility | More availability in dosing options. | Limited dosing options. |
| Drug interactions | No known drug interactions. | Many drug-drug interactions are known for the oral formulation. |
| Mechanism of action |
|---|
| Agonists5,8-13 |
| Work by overstimulating the LHRH (also known as GnRH) receptors in the anterior pituitary gland, which desensitizes them over time. Continual binding of the agonists to the LHRH receptors results in an initial increase in LH release and T production, followed by a steep drop in both. Patients will reach clinically castrate levels of T in 2 to 4 weeks. |
| Antagonists5,8-13 |
| Work by blocking the GnRH (gonadotropin-releasing hormone) receptors in the pituitary gland, causing a faster decrease in LH and T, with castrate levels being reached in 2 to 3 days. |
| Benefits of initiating |
|---|
| Agonists5,8-13 |
| Long-acting formulations provide patients with reliable, controlled suppression over time, which may help avoid periods of T breakthrough above castrate levels. |
| Antagonists5,8-13 |
| May benefit patients with spinal metastases due to the absence of initial T increase and others who require a rapid T suppression. |
| Route of delivery |
|---|
| Agonists5,8-13 |
| Injected subcutaneously or intramuscularly. |
| Antagonists5-11 |
| Subcutaneous injection or oral. |
| Administration schedule |
|---|
| Agonists5,8-13 |
| Depending on the product, can be administered monthly, every 3 or 4 months, or even just twice per year. |
| Antagonists5-11 |
| Taken once a day orally or injected every month. |
| Drug interactions |
|---|
| Agonists5,8-13 |
| No known drug interactions. |
| Antagonists5,8-13 |
| Many drug-drug interactions are known for the oral formulation. |
| aPC stage | How to determine prognosis | Treatments available |
|---|---|---|
| Biochemical recurrence2,14 |
Counsel patients of the risk of developing metastatic disease. Continue PSA measurements and clinical evaluations. Consider radiographic assessments based on overall PSA and PSA kinetics. If PSADT <12 months, perform periodic staging evaluations consisting of cross-sectional imaging such as CT or MRI and technetium bone scan and/or PSMA-PET. |
If no metastatic disease is shown on imaging, observe or offer clinical trial enrollment. ADT and enzalutamide for patients with high-risk biochemical recurrence. |
| mCSPC2 |
Assess the extent of metastatic disease (low vs high volume)—high-volume is defined as ≥4 bone metastases with at least one metastasis outside of the spine/pelvis and/or the presence of visceral metastases. Assess if newly diagnosed mCSPC patient is experiencing symptoms from metastatic disease at time of presentation to guide discussions of prognosis and further disease management. Obtain baseline PSA and serial PSA at 3-to-6-month intervals after initiation of ADT and consider periodic conventional imaging. Offer genetic testing. |
ADT with GnRH agonists or antagonists or surgical castration. ADT in combination with either ARPI (abiraterone acetate plus prednisone, apalutamide, enzalutamide) or chemotherapy (docetaxel). In patients with de novo mCSPC, ADT in combination with docetaxel and either abiraterone acetate plus prednisone or darolutamide. In mCSPC patients with low-volume metastatic disease, primary radiotherapy to the prostate in combination with ADT. Do not offer bicalutamide, flutamide, or nilutamide in combination with GnRH agonists, except to block T flares. Do not offer oral ARPI without ADT. |
| nmCRPC2 |
Obtain serial PSA measurements at 3-to-6-month intervals. Calculate a PSADT starting at the time of development of castration resistance. Assess patients for development of metastatic disease using conventional or PSMA-PET imaging at intervals of 6-to-12 months. |
Apalutamide, darolutamide, and enzalutamide with continued ADT for those at high risk for developing metastatic disease (PSADT ≤10 months). Observe those with continued ADT who are at lower risk (PSADT >10 months) for developing metastatic disease. Do not offer systemic chemotherapy or immunotherapy outside of a clinical trial. |
| mCRPC2,15,16 |
Obtain baseline labs (eg, PSA, testosterone, LDH, Hgb, alkaline phosphatase level) and review location of metastatic disease (lymph node, bone, visceral), disease-related symptoms, and PS to inform discussions of prognosis and treatment decision-making. In mCRPC patients without PSA progression or new symptoms, perform imaging at least annually. In mCRPC patients with disease progression (PSA or radiographic progression or new disease-related symptoms) having previously received docetaxel and androgen pathway inhibitor, who are considering 177Lu-PSMA-617, order PSMA-PET imaging. Offer genetic testing. |
In newly diagnosed mCRPC patients who have not received prior ARPIs, continue ADT with abiraterone acetate plus prednisone, docetaxel, or enzalutamide.
Sipuleucel-T before chemotherapy in patients who are asymptomatic or minimally symptomatic. Radium-223 for patients with painful bone metastases and without known visceral disease or lymphadenopathy. 177Lu-PSMA-617 in patients who have received docetaxel and ARPI and have cancer visible on PSMA-PET. Taxane chemotherapy given with prednisone (docetaxel and cabazitaxel).
PARP inhibitors for patients with deleterious or suspected deleterious germline or somatic HRR gene-mutated mCRPC following prior treatment with enzalutamide or abiraterone acetate and/or a taxane-based chemotherapy.
For patients with MMR deficient or MSI-H mCRPC, pembrolizumab. PSMA radionuclide therapy. |
| aPC stage - Biochemical recurrence2,4 |
|---|
| How to determine prognosis |
| Counsel patients of the risk of developing metastatic disease.
|
| Treatments available |
| If no metastatic disease is shown on imaging, observe or offer clinical trial enrollment.
|
| aPC stage - mCSPC2 |
|---|
| How to determine prognosis |
| Assess the extent of metastatic disease (low vs high volume)—high-volume is defined as ≥4 bone metastases with at least one metastasis outside of the spine/pelvis and/or the presence of visceral metastases.
|
| Treatments available |
| ADT with GnRH agonists or antagonists or surgical castration.
|
| aPC stage - nmCRPC2 |
|---|
| How to determine prognosis |
| Obtain serial PSA measurements at 3-to-6-month intervals.
|
| Treatments available |
| Apalutamide, darolutamide, and enzalutamide with continued ADT for those at high risk for developing metastatic disease (PSADT ≤10 months).
|
| aPC stage - mCRPC2,15,16 |
|---|
| How to determine prognosis |
| Obtain baseline labs (eg, PSA, testosterone, LDH, Hgb, alkaline phosphatase level) and review location of metastatic disease (lymph node, bone, visceral), disease-related symptoms, and PS to inform discussions of prognosis and treatment decision-making.
|
| Treatments available |
In newly diagnosed mCRPC patients who have not received prior ARPIs, continue ADT with abiraterone acetate plus prednisone, docetaxel, or enzalutamide.
Sipuleucel-T before chemotherapy in patients who are asymptomatic or minimally symptomatic. Radium-223 for patients with painful bone metastases and without known visceral disease or lymphadenopathy. Lutetium-PSMA-617 (Lu-PSMA-617) in patients who have received docetaxel and ARPI and have cancer visible on PSMA-PET. Taxane chemotherapy given with prednisone (docetaxel and cabazitaxel).
PARP inhibitors for patients with deleterious or suspected deleterious germline or somatic HRR gene-mutated mCRPC following prior treatment with enzalutamide or abiraterone acetate and/or a taxane-based chemotherapy.
For patients with MMR deficient or MSI-H mCRPC, pembrolizumab. PSMA radionuclide therapy. s. |
Advanced prostate cancer treatment can be complex and cause numerous side effects. It’s important to provide patients with comprehensive care by ensuring they have coordinated care between a team of physicians such as urologists, oncologists, primary care physicians, cardiologists, dietitians, physical therapists, social workers, psychologists, and more.2
Working directly as a multidisciplinary team allows elevated care for patients. Having trusted multidisciplinary HCPs available during treatment can allow you firsthand insight into how the patient’s disease is progressing, how treatment is working, and what side effects they need assistance with.
The Prostate Cancer 360 (PC360) initiative was created by a team of 14 interdisciplinary experts who have a dedicated clinical interest in prostate cancer and ADT management. The group consists of17:
These experts worked together to define challenges associated with ADT adverse event management and developed recommendations for ADT prescribers so they can provide better care to patients.17
Since ADT is a foundational therapy for advanced prostate cancer, the PC360 recommendations should be considered during continued treatment for aPC.
Learn more about PC360 at ProstateCancer360.com.